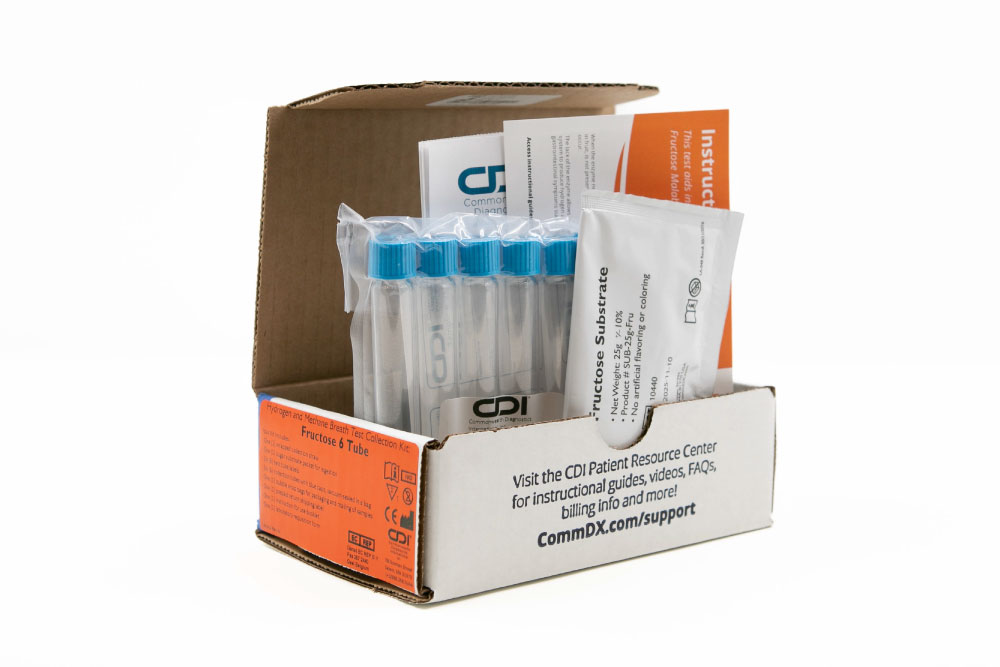
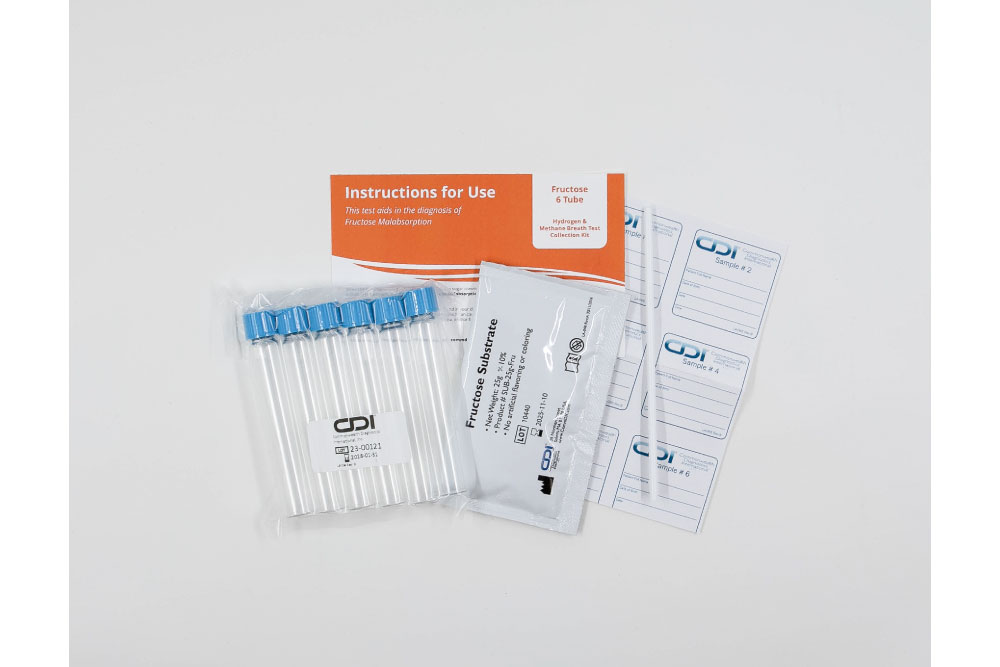
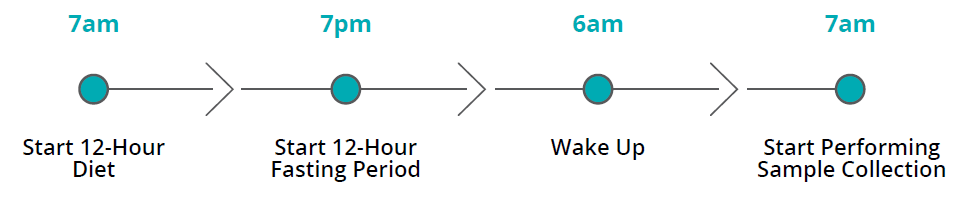
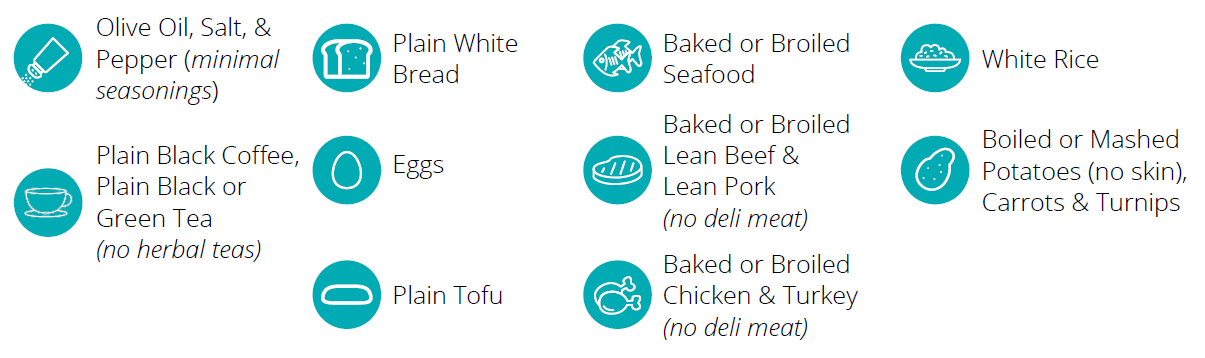
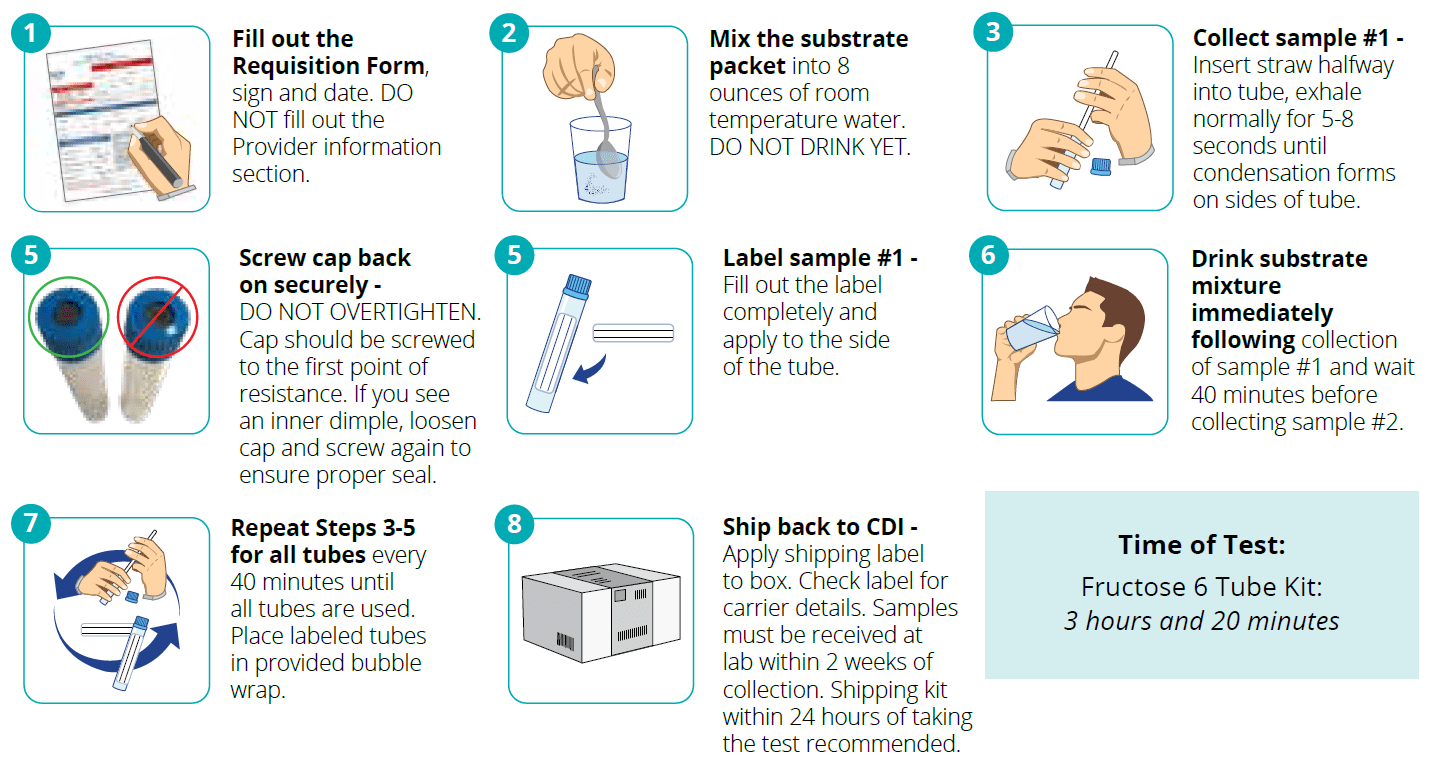
Indications and Usage: The intended use of this device is for the collection of human breath samples to aid in the diagnosis of fructose malabsorption.
Contraindications: Patients with a known fructose allergy should not take this test. Patients with a low galactose diet should discuss with their healthcare provider prior to taking this test. Patients with diabetes should not use this test due to the amount of absorbable sugar and fasting required. Patients should discuss with their healthcare provider prior to ingesting the substrate.
Precautions: Patients with food allergies should take precaution before taking this test as most substrates do not come with food allergy labeling. Contact immediate medical assistance if you have signs of a possible allergic reaction: hives; difficulty breathing; swelling of face, lips, tongue, and throat. Any serious incident that occurs in relation to this device shall be reported to the manufacturer and the competent authority of the member state in which the patient is established.
Please consult with your healthcare provider if you have any health concerns, are on a specialty diet, and/or are on prescribed medications. Please do not discontinue taking any prescribed or over-the-counter (OTC) medications, including the ones listed in the Test Restrictions section, without consulting with your healthcare provider prior to taking this test.
* The substrate is provided “as is” and CDI makes no representations or warranties whatsoever, express or implied, including without limitation any implied warranty of merchantability or fitness for a particular purpose. Patients should consult with their healthcare provider before ingesting this substrate.